Chemical Reactions and Equations - Class 10th Science
Oxidation And Reduction
Oxidation
Gain of oxygen or loss of hydrogen by an atom or compound or molecule or ion is called oxidation. And the reaction takes place in this process is called Oxidation Reaction.
Oxidizing Agent
Atom, compound or ion which gives oxygen to another substance or removes hydrogen from a substance is called oxidizing agent.
Example: 1:
Oxidation because of gains of oxygen
When copper is heated in air, it gets oxidized and gives copper oxide.
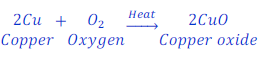
In this reaction copper gains oxygen, and hence copper becomes oxidized. Reaction is called oxidation reaction.
In this reaction, oxygen gives oxygen to copper, hence oxygen is here oxidizing agent.
Example: 2:
When magnesium ribbon is burnt in the presence of air, magnesium gets oxidized and gives magnesium oxide.
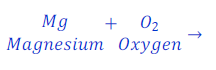
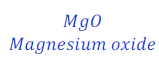
In this reaction, magnesium gains oxygen from air, and hence gets oxidized. This is a oxidation reaction.
In this reaction, oxygen gives oxygen to magnesium, hence oxygen is here oxidizing agent.
Example: 3:
Oxidation because of losing of hydrogen
When natural gas is burnt in air, it gives carbon dioxide.
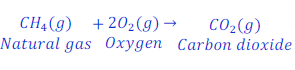
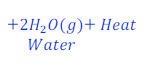
In this reaction, natural gas loses hydrogen and gives carbon dioxide. Since, carbon present in natural gas loses hydrogen thus, carbon gets oxidized here.
Here, oxygen, removes hydrogen from natural gas(methane), thus oxygen is here oxidizing agent.
Example: 4:
When methane reacts with chlorine, it loses hydrogen and gives methyl chloride.
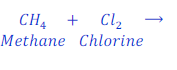
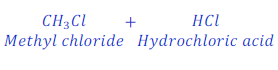
In this reaction oxidation of carbon present in methane or methane takes place because of loss of hydrogen. Carbon present in methane or methane itself loses hydrogen and forms methyl chloride because of oxidation.
In this reaction, chlorine removes hydrogen from methane (natural gas), thus, chlorine is oxidizing agent.
Reduction
Loss of oxygen or gain of hydrogen by an atom or compound or molecule or ion is called reduction. And the reaction takes place in this process is called Reduction Reaction.
Reducing Agent
Atom, compound or ion which removes oxygen from another substance or gives hydrogen from a substance is called reducing agent.
Example: 1:
Reduction because of loss of oxygen
When copper oxide is heated with hydrogen, it loses oxygen and is reduced to copper.
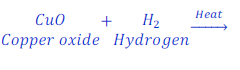
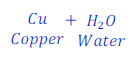
In this reaction, copper oxide loses oxygen, and hence is reduced to copper.
Here, since, hydrogen reduces oxygen from copper oxide, thus hydrogen is a reducing agent.
Example: 2:
Reduction because of gain of hydrogen
When chlorine reacts with hydrogen, it gains hydrogen and reduced to hydrochloric acid.
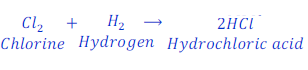
In this reaction, chlorine gains hydrogen and gets reduced to hydrochloric acid.
In this reaction, hydrogen is reducing agent, as it gives hydrogen to chlorine molecule.